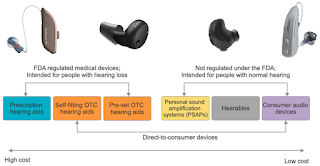
While hearing aids are the most common treatment for age-related hearing loss, low uptake of hearing aids is due to high cost, stigma, and a lack of perceived need. To increase accessibility and affordability, the U.S. Food and Drug Administration created a new OTC hearing aid category. Currently, there are various hearing devices available for individuals with and without hearing loss, including medical devices (prescription hearing aids, self-fitting OTC hearing aids, and pre-set OTC hearing aids) and non-medical devices (PSAPs, hearables, and consumer audio devices such as Apple's AirPods Pro and Live Listen feature, Samsung's Galaxy Buds and Hearing Aid feature, and Bose Hearphones).
Regulated by the FDA, hearing aids have evolved rapidly in the last decade, with features, functionalities, and designs improving significantly. However, there is a lack of research on all aspects of OTC hearing aids currently on the market. High-quality independent research is necessary to supplement evidence provided by OTC hearing aid manufacturers for regulatory approval purposes. Recently published article has reviewed existing research on direct-to-consumer (DTC) hearing devices such as PSAPs and highlighted the need for immediate research on OTC hearing aids and service delivery models to inform policy and clinical care.
REFERENCE
Manchaiah V, Swanepoel W, Sharma A. Prioritizing research on over-the-counter (OTC) hearing aids for age-related hearing loss. Front Aging. 2023 Mar 23;4:1105879. doi: 10.3389/fragi.2023.1105879. PMID: 37033402; PMCID: PMC10078955.
No comments:
Post a Comment